Obesity and Aging: How Excess Weight Steals Your Youth
This article explains how obesity accelerates the biological aging process at a cellular and systemic level, leading to premature health decline and chronic diseases.
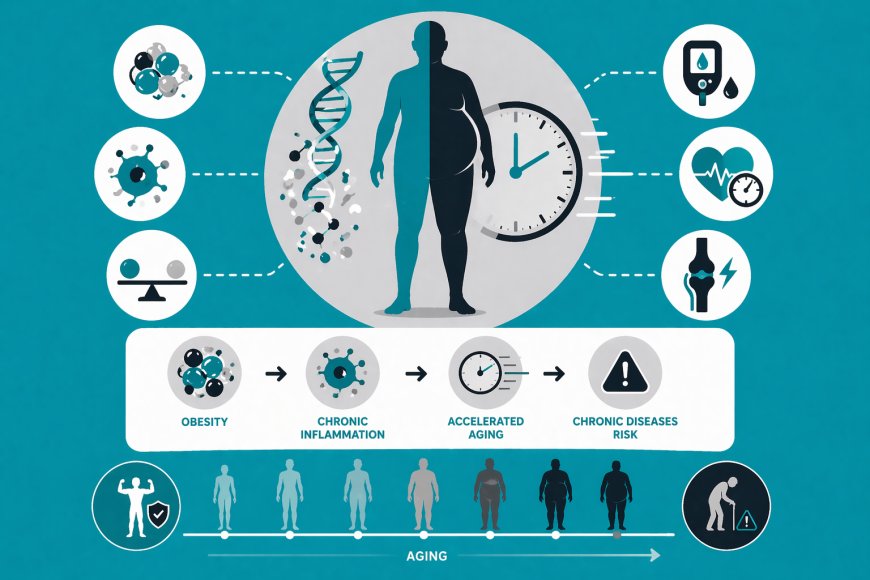
Aging has long been associated with the passage of time, yet modern science reveals a more complex reality: biological age does not always align with chronological age. Individuals in midlife may exhibit physiological characteristics of much older individuals, not solely due to genetics, but as a result of accumulated metabolic and environmental factors. Among these, obesity stands as a central driver, accelerating aging across cellular, functional, and structural systems.
Obesity as a silent driver of biological decline
The biological footprint of obesity reflects a cascade of cumulative effects that influence nearly every system in the body. One of the earliest manifestations appears at the tissue level, where visceral fat accumulation promotes chronic low-grade inflammation. This persistent inflammatory state contributes to progressive tissue damage.
The liver, for example, becomes vulnerable to non-alcoholic fatty liver disease, gradually impairing its function and structure. Blood vessels lose elasticity due to lipid deposition, leading to vascular stiffness and reduced circulation. These internal changes reflect a form of aging that is not immediately visible, yet profoundly impactful.
Cellular aging and functional deterioration
At the cellular level, obesity accelerates cellular senescence, a state in which cells lose their capacity to divide and regenerate. These senescent cells release inflammatory signals that disrupt surrounding tissues and weaken immune function.
This process increases susceptibility to chronic diseases such as diabetes, cancer, and neurodegenerative disorders. It also reduces the body's ability to repair itself, reinforcing a cycle of decline.
Premature physical aging
Obesity imposes a mechanical and metabolic burden on the body. Individuals often experience reduced mobility, diminished cardiovascular efficiency, and persistent fatigue. Over time, decreased physical activity leads to muscle atrophy, creating a feedback loop of weakness and inactivity.
This pattern mirrors the physical decline typically associated with advanced age, even when chronological age remains relatively young.
Cognitive and psychological impact
The effects of obesity extend into cognitive function. Research links obesity with slower mental processing, reduced memory performance, and increased risk of dementia.
These outcomes are driven by multiple mechanisms, including impaired cerebral blood flow, neuroinflammation, and hormonal imbalance. The result is a decline in cognitive resilience and adaptability.
Obesity and chronic disease interactions
Hypertension and fluid imbalance
Chronic high blood pressure places continuous stress on blood vessel walls, reducing their elasticity and increasing permeability. This leads to fluid leakage into surrounding tissues, resulting in edema.
Fluid accumulation in extremities causes swelling and reduced mobility, while in advanced cases, fluid may accumulate in the lungs, impairing breathing and contributing to persistent fatigue. This physiological burden resembles advanced aging, occurring prematurely.
In severe cases, fluid imbalance may extend to the brain, contributing to increased intracranial pressure and gradual cognitive decline.
Diabetes and systemic deterioration
Type 2 diabetes, closely linked to obesity, accelerates aging through damage to nerves and vision. Chronic hyperglycemia affects small blood vessels, leading to diabetic neuropathy.
This condition begins with mild sensory changes and progresses to loss of sensation and impaired motor responses. It also affects autonomic functions such as heart rate and digestion, creating systemic instability.
In the retina, vascular damage leads to diabetic retinopathy, a major cause of vision loss. As these impairments progress, individuals experience reduced independence, similar to advanced aging.
Insulin resistance and brain aging
Insulin resistance extends beyond metabolic disruption to directly affect brain function. Insulin plays a role in neuronal communication, memory formation, and cognitive flexibility.
When this pathway is impaired, cognitive performance declines. Brain regions associated with memory, such as the hippocampus, are particularly affected. This overlap has led some researchers to describe Alzheimer’s disease as a form of metabolic dysfunction linked to insulin resistance.
Chronic neuroinflammation further accelerates cognitive decline, weakening the brain’s protective barriers and increasing vulnerability to damage.
Conclusion
Obesity is not merely an excess of body weight, but a complex metabolic condition that accelerates biological aging across multiple systems. Its effects manifest in tissue damage, cellular dysfunction, physical decline, and cognitive impairment.
It operates silently, often without immediate symptoms, yet progressively alters the body’s internal timeline. Addressing obesity requires more than aesthetic consideration; it demands recognition as a central determinant of long-term health, functional independence, and biological age.
The challenge is not only to reduce weight, but to restore physiological balance and slow the accelerated passage of time within the body.
What's Your Reaction?